Minerals
Yes.
Elements
Pure substance that cannot be separated.
Atom
Smallest piece of element containing basic characteristic: protons + neutrons.
Ion
Non-netural atoms (anion—extra negative charge, more electrons that protons; cation—extra positive charge, less electrons than protons).
Bonds
Bonds forms. Arrange of bonds can determine mineral properties (diamond vs. graphite).
Ionic
When two polarized atoms attract: cation and anions attract
Covalent
When atoms share electrons
Polymorph
Minerals with the same composition but different structures like diamond (hardest material) and graphite (softest.)
Forming minerals
New Minerals mostly from from a solution (magma, seawater, etc.) using some geological process: freezing, precipitation, chemical reaction, biogenic (shells.)
Sometimes, Minerals form in place from metamorphism.
Early crystals act as a seed for future mineral growth, esp., when crystals can grow into open space.
Classifying Minerals
Classified by dominant anion.
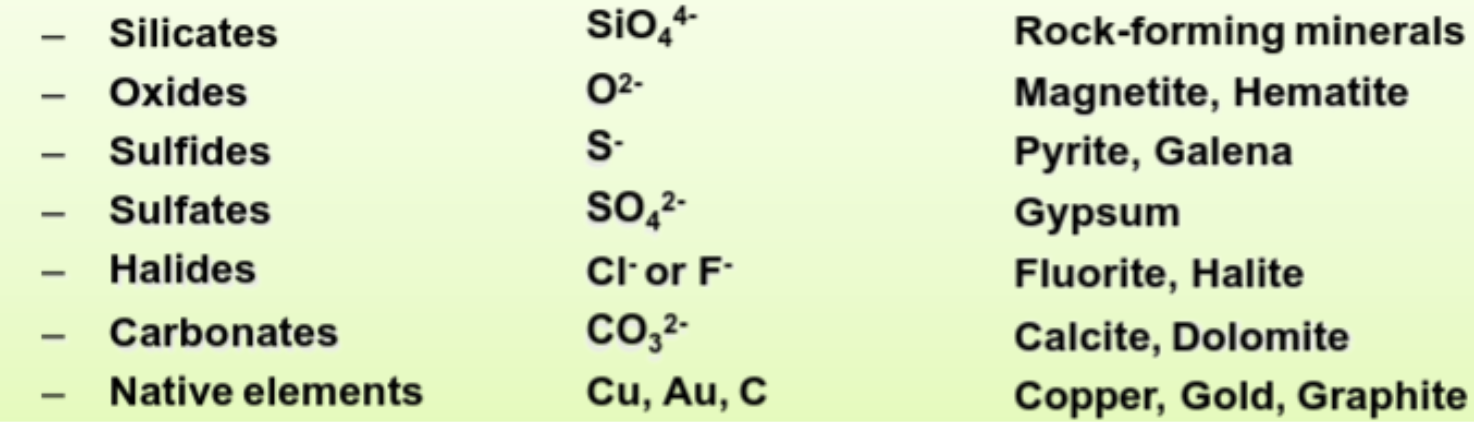
Silicates
Most important minerals: silicates. “Rock forming minerals.” 95% of continental crust. \(SiO_{4}^{4-}\) anion. They are bonded together by cations. How they bond determines structure.
single-chain sillicates
Two of the three basal oxygens (base plane of the coordinating polyhedron) bounded together.
sheet sillicates
All three basal oxygens bonded
framework sillicates
All oxygen between adjacent tetrhedra, basal or not, are shared
Classifying Minerals
4kish materials, some words to talk about them:
- color: “color, raw specimen”,, can help malachite but can’t be used to help quarts
- luster: how the material scatters light
- hardness: scratching resistance of a material, which is a function of atomic bond stretch; talc the weakest diamond the strongest
- streak: “color, powdered material”, what the material leaves in terms color of crushed powder on unglazed porcelain plate
- cleavage: materials tend to break along lattice planes; when there are not weak bonds, then the material fractures instead of cleaves
- specific gravity: material weight over weight of equal water
- reaction to acid
- crystal habit: the shape that crystals tend to grow in
- fracture tendency: how easily the material fractures
Mostly wild rocks can’t be really identified by normal sampling; rock type and tectonic setting helps more.
Electron microscope can be gold standard but expensive.
Mineral vs Rock
Rock are made of minerals.
Most rocks contain more than one mineral but some are monomineraliic like limestone (calcite), rock salt (halite), or ice.